药品详细
Prednisolone(泼尼松龙)
化学结构式图
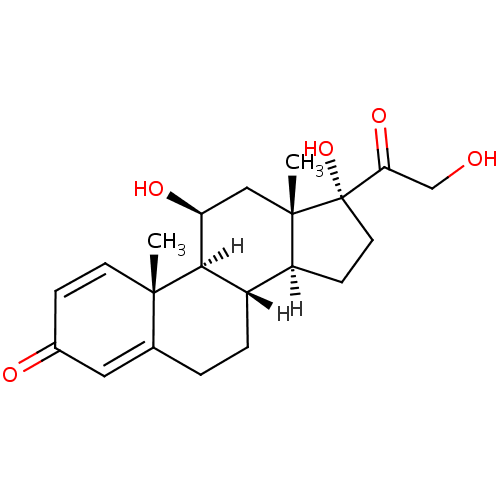
中文名
泼尼松龙
英文名
Prednisolone
分子式
C21H28O5
化学名
(1S,2R,10S,11S,14R,15S,17S)-14,17-dihydroxy-14-(2-hydroxyacetyl)-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadeca-3,6-dien-5-one
分子量
Average: 360.444
Monoisotopic: 360.193674006
Monoisotopic: 360.193674006
CAS号
50-24-8
ATC分类
A07E Intestinal Antiinflammatory Agents;C05A 未知;D07A 未知;D07A 未知;D07A 未知;D07X 未知;D10A Anti-Acne Preparations for Topical Use;H02A 未知;H02A 未知;R01A 未知;S01B 抗炎药;S01C 炎药组合;S02B 未知;S03B 未知;H02A 未知
药物类型
small molecule
阶段
approved
商品名
同义名
基本介绍
A glucocorticoid with the general properties of the corticosteroids. It is the drug of choice for all conditions in which routine systemic corticosteroid therapy is indicated, except adrenal deficiency states. [PubChem]
生产厂家
- Akorn inc
- Alcon laboratories inc
- Alcon universal ltd
- Allergan pharmaceutical
- Alpharma uspd inc
- Amneal pharmaceuticals
- Apotex inc richmond hill
- Barr laboratories inc
- Bausch and lomb pharmaceuticals inc
- Bel mar laboratories inc
- Central pharmaceuticals inc
- Cm bundy co
- Elkins sinn div ah robins co inc
- Everylife
- Ferndale laboratories inc
- Halsey drug co inc
- Heather drug co inc
- Hi tech pharmacal co inc
- Impax laboratories inc
- Inwood laboratories inc sub forest laboratories inc
- Ivax pharmaceuticals inc sub teva pharmaceuticals usa
- John j ferrante
- Kv pharmaceutical co
- L perrigo co
- Lannett co inc
- Marshall pharmacal corp
- Merck and co inc
- Muro pharmaceutical inc
- Novartis pharmaceuticals corp
- Paddock laboratories inc
- Panray corp sub ormont drug and chemical co inc
- Pfizer laboratories div pfizer inc
- Pharmaceutical assoc inc
- Pharmaceutical assoc inc div beach products
- Pharmacia and upjohn co
- Pharmafair inc
- Phoenix laboratories inc
- Private formulations inc
- Purepac pharmaceutical co
- Roxane laboratories inc
- Sandoz canada inc
- Sandoz inc
- Schering corp sub schering plough corp
- Shionogi pharma inc
- Sola barnes hind
- Sperti drug products inc
- Superpharm corp
- Tablicaps inc
- Taro pharmaceuticals usa inc
- Teva parenteral medicines inc
- Teva pharmaceuticals usa
- Teva pharmaceuticals usa inc
- Ucb inc
- Udl laboratories inc
- Valeant pharmaceuticals international
- Vintage pharmaceuticals llc
- Vitarine pharmaceuticals inc
- Watson laboratories inc
- We pharmaceuticals inc
- West ward pharmaceutical corp
- Whiteworth towne paulsen inc
- Wockhardt eu operations (swiss) ag
封装厂家
- Adamis Laboratories
- Aidarex Pharmacuticals LLC
- Akorn Inc.
- Alcon Laboratories
- Allergan Inc.
- Amend
- Amneal Pharmaceuticals
- A-S Medication Solutions LLC
- Atlantic Biologicals Corporation
- Bausch & Lomb Inc.
- Bedford Labs
- Ben Venue Laboratories Inc.
- BioMarin Pharmaceuticals Inc.
- Blansett Pharmacal Co. Inc.
- Bryant Ranch Prepack
- C.O. Truxton Inc.
- Cardinal Health
- Carlisle Laboratories Inc.
- Cima Laboratories Inc.
- Clint Pharmaceutical Inc.
- Consolidated Midland Corp.
- Darby Dental Supply Co. Inc.
- Dexo LLC
- Dispensing Solutions
- Diversified Healthcare Services Inc.
- Ethex Corp.
- Falcon Pharmaceuticals Ltd.
- Hawthorn Pharmaceuticals
- Hi Tech Pharmacal Co. Inc.
- Keene Pharmaceuticals Inc.
- KV Pharmaceutical Co.
- Lake Erie Medical and Surgical Supply
- Laser Pharmaceuticals
- Lunsco Inc.
- Lyne Laboratories Inc.
- Major Pharmaceuticals
- Marpac
- Martica Enterprises Inc.
- Martin Surgical Supply
- Medisca Inc.
- Merit Pharmaceuticals
- MWI Veterinary Supply Co.
- National Pharmaceuticals
- Novartis AG
- Ocusoft
- OMJ Pharmaceuticals
- Pacific Pharma Lp
- Paddock Labs
- Palmetto Pharmaceuticals Inc.
- Pfizer Inc.
- Pharmaceutical Association
- Pharmacia Inc.
- Pharmedix
- Physicians Total Care Inc.
- Preferred Pharmaceuticals Inc.
- Prescript Pharmaceuticals
- Primedics Laboratories
- Qualitest
- Rebel Distributors Corp.
- San Jose Surgical Supply Inc.
- Sandoz
- Sciele Pharma Inc.
- Shionogi Pharma Inc.
- Stat Rx Usa
- Taro Pharmaceuticals USA
- Teva Pharmaceutical Industries Ltd.
- UCB Pharma
- Upstate Pharma LLC
- Vedco Inc.
- Vintage Pharmaceuticals Inc.
- Wa Butler Co.
- Watson Pharmaceuticals
- Wockhardt Ltd.
参考
| Synthesis Reference | Not Available |
| General Reference | Not Available |
剂型
规格
化合物类型
| Type | small molecule |
| Classes |
|
| Substructures |
|
适应症
药理
| Indication | For the treatment of primary or secondary adrenocortical insufficiency, such as congenital adrenal hyperplasia, thyroiditis. Also used to treat psoriatic arthritis, rheumatoid arthritis, ankylosing spondylitis, bursitis, acute gouty arthritis and epicondylitis. Also indicated for treatment of systemic lupus erythematosus, pemphigus and acute rhematic carditis. Can be used in the treatment of leukemias, lymphomas, thrombocytopenia purpura and autoimmune hemolytic anemia. Can be used to treat celiac disease, insulin resistance, ulcerative colitis and liver disorders. | |||||||||
| Pharmacodynamics | Prednisolone is a synthetic glucocorticoid used as antiinflammatory or immunosuppressive agent. Prednisolone is indicated in the treatment of various conditions, including congenital adrenal hyperplasia, psoriatic arthritis, systemic lupus erythematosus, bullous dermatitis herpetiformis, seasonal or perennial allergic rhinitis, allergic corneal marginal ulcers, symptomatic sarcoidosis, idiopathic thrombocytopenic purpura in adults, leukemias and lymphomas in adults, and ulcerative colitis. Glucocorticoids are adrenocortical steroids and cause profound and varied metabolic effects. In addition, they modify the body's immune responses to diverse stimuli. | |||||||||
| Mechanism of action | Glucocorticoids such as Prednisolone can inhibit leukocyte infiltration at the site of inflammation, interfere with mediators of inflammatory response, and suppress humoral immune responses. The antiinflammatory actions of glucocorticoids are thought to involve phospholipase A2 inhibitory proteins, lipocortins, which control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes. Prednisolone reduces inflammatory reaction by limiting the capillary dilatation and permeability of the vascular structures. These compounds restrict the accumulation of polymorphonuclear leukocytes and macrophages and reduce the release of vasoactive kinins. Recent research suggests that corticosteroids may inhibit the release of arachidonic acid from phospholipids, thereby reducing the formation of prostaglandins. Prednisolone is a glucocorticoid receptor agonist. On binding, the corticoreceptor-ligand complex translocates itself into the cell nucleus, where it binds to many glucocorticoid response elements (GRE) in the promoter region of the target genes. The DNA bound receptor then interacts with basic transcription factors, causing an increase or decrease in expression of specific target genes, including suppression of IL2 (interleukin 2) expression. | |||||||||
| Absorption | Readily absorbed by gastrointestinal tract, peak plasma concentration is reached 1-2 hours after administration. | |||||||||
| Volume of distribution | Not Available | |||||||||
| Protein binding | Very high (>90%) | |||||||||
| Metabolism |
Excreted in the urine as either free or glucoconjugate.
|
|||||||||
| Route of elimination | Not Available | |||||||||
| Half life | 2-3 hours | |||||||||
| Clearance | Not Available | |||||||||
| Toxicity | LD50=500 mg/kg (oral, rat), short-term side effects include high blood glucose levels and fluid retention. Long term side effects include Cushing's syndrome, weight gain, osteoporosis, glaucoma, type II diabetes and adrenal suppression. | |||||||||
| Affected organisms |
|
|||||||||
| Pathways |
|
理化性质
| Properties | |||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| State | solid | ||||||||||||||||||||||||||||||||||||||||||
| Experimental Properties |
|
||||||||||||||||||||||||||||||||||||||||||
| Predicted Properties |
|
||||||||||||||||||||||||||||||||||||||||||
药物相互作用
| Drug | Interaction |
|---|---|
| Acenocoumarol | The corticosteroid, prednisolone, alters the anticoagulant effect, acenocoumarol. |
| Acetylsalicylic acid | The corticosteroid, prednisolone, may decrease the effect of the salicylate, acetylsalicylic acid. |
| Ambenonium | The corticosteroid, prednisolone, may decrease the effect of the anticholinesterase, ambenonium. |
| Amobarbital | The barbiturate, amobarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Anisindione | The corticosteroid, prednisolone, alters the anticoagulant effect of anisindione. |
| Aprobarbital | The barbiturate, aprobarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Bismuth Subsalicylate | The corticosteroid, prednisolone, may decrease the effect of the salicylate, bismuth subsalicylate. |
| Butabarbital | The barbiturate, butabarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Butalbital | The barbiturate, butalbital, may decrease the effect of the corticosteroid, prednisolone. |
| Butethal | The barbiturate, butethal, may decrease the effect of the corticosteroid, prednisolone. |
| Chlorotrianisene | The estrogenic agent, chlorotrianisene, may increase the effect of the corticosteroid, prednisolone. |
| Clomifene | The estrogenic agent, clomifene, may increase the effect of the corticosteroid, prednisolone. |
| Conjugated Estrogens | The estrogenic agent may increase the effect of the corticosteroid, prednisolone. |
| Dicumarol | The corticosteroid, prednisolone, alters the anticoagulant effect of dicumarol. |
| Diethylstilbestrol | The estrogenic agent, diethylstilbestrol, may increase the effect of the corticosteroid, prednisolone. |
| Dihydroquinidine barbiturate | The barbiturate, dihydroquinidine barbiturate, may decrease the effect of the corticosteroid, prednisolone. |
| Edrophonium | The corticosteroid, prednisolone, may decrease the effect of the anticholinesterase, edrophonium. |
| Estradiol | The estrogenic agent, estradiol, may increase the effect of the corticosteroid, prednisolone. |
| Estriol | The estrogenic agent, estriol, may increase the effect of the corticosteroid, prednisolone. |
| Estrone | The estrogenic agent, estrone, may increase the effect of the corticosteroid, prednisolone. |
| Estropipate | The estrogenic agent, estropipate, may increase the effect of the corticosteroid, prednisolone. |
| Ethinyl Estradiol | The estrogenic agent, ethinyl estradiol, may increase the effect of the corticosteroid, prednisolone. |
| Ethotoin | The enzyme inducer, ethotoin, may decrease the effect of the corticosteroid, prednisolone. |
| Fosphenytoin | The enzyme inducer, fosphenytoin, may decrease the effect of the corticosteroid, prednisolone. |
| Heptabarbital | The barbiturate, heptabarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Hexobarbital | The barbiturate, hexobarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Itraconazole | The imidazole, itraconazole, may increase the effect and toxicity of the corticosteroid, prednisolone. |
| Ketoconazole | The imidazole, ketoconazole, may increase the effect and toxicity of the corticosteroid, prednisolone. |
| Magnesium salicylate | The corticosteroid, prednisolone, may decrease the effect of magnesium salicylate. |
| Mephenytoin | The enzyme inducer, mephenytoin, may decrease the effect of the corticosteroid, prednisolone. |
| Mestranol | The estrogenic agent, mestranol, may increase the effect of the corticosteroid, prednisolone. |
| Methohexital | The barbiturate, methohexital, may decrease the effect of the corticosteroid, prednisolone. |
| Methylphenobarbital | The barbiturate, methylphenobarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Midodrine | Increased arterial pressure |
| Neostigmine | The corticosteroid, prednisolone, may decrease the effect of the anticholinesterase, neostigmine. |
| Pentobarbital | The barbiturate, pentobarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Phenobarbital | The barbiturate, phenobarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Phenytoin | The enzyme inducer, phenytoin, may decrease the effect of the corticosteroid, prednisolone. |
| Primidone | The barbiturate, primidone, may decrease the effect of the corticosteroid, prednisolone. |
| Pyridostigmine | The corticosteroid, prednisolone, may decrease the effect of the anticholinesterase, pyridostigmine. |
| Quinestrol | The estrogenic agent, quinestrol, may increase the effect of the corticosteroid, prednisolone. |
| Quinidine barbiturate | The barbiturate, quinidine barbiturate, may decrease the effect of the corticosteroid, prednisolone. |
| Rifampin | The enzyme inducer, rifampin, may decrease the effect of the corticosteroid, prednisolone. |
| Salicylate-sodium | The corticosteroid, prednisolone, may decrease the effect of the salicylate, salicylate-sodium. |
| Salsalate | The corticosteroid, prednisolone, may decrease the effect of the salicylate, salsalate. |
| Secobarbital | The barbiturate, secobarbital, may decrease the effect of the corticosteroid, prednisolone. |
| Tacrine | Tacrine and Prednisolone may independently exacerbate muscle weakness in myasthenia gravis patients. Monitor for additive muscle weakness effects. |
| Talbutal | The barbiturate, talbutal, may decrease the effect of the corticosteroid, prednisolone. |
| Trastuzumab | Trastuzumab may increase the risk of neutropenia and anemia. Monitor closely for signs and symptoms of adverse events. |
| Trisalicylate-choline | The corticosteroid, prednisolone, may decrease the effect of the salicylate, trisalicylate-choline. |
| Vecuronium | Vecuronium may increase the adverse neuromuscular effects of systemic corticosteroids, such as Prednisolone. Monitor for increased muscle weakness and signs of polyneuropathies and myopathy. |
| Warfarin | The corticosteroid, prednisolone, alters the anticoagulant effect of warfarin. |
食物相互作用
- Avoid alcohol. Avoid caffeine.
- Take with food to reduce gastric irritation.

