药品详细
Etoposide (依托泊苷 )
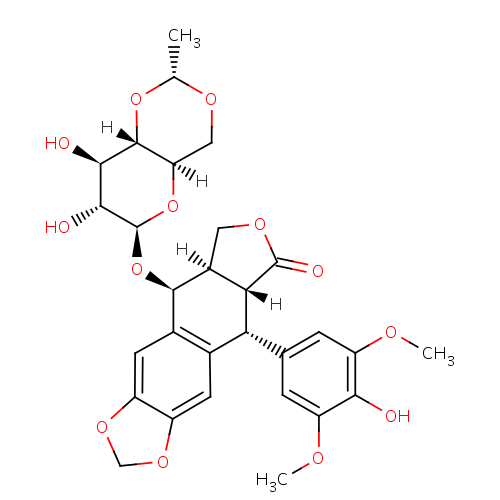
化学结构式图
中文名
依托泊苷
英文名
Etoposide
分子式
Not Available
化学名
(10R,11R,15R,16S)-16-{[(2R,4aR,6R,7R,8R,8aS)-7,8-dihydroxy-2-methyl-hexahydro-2H-pyrano[3,2-d][1,3]dioxin-6-yl]oxy}-10-(4-hydroxy-3,5-dimethoxyphenyl)-4,6,13-trioxatetracyclo[7.7.0.0^{3,7}.0^{11,15}]hexadeca-1(9),2,7-trien-12-one
分子量
Average: 588.5566
Monoisotopic: 588.184291110
Monoisotopic: 588.184291110
CAS号
33419-42-0
ATC分类
L01C 植物碱和其他天然产物
药物类型
small molecule
阶段
商品名
Eposin;Etopophos;Lastet;Toposar;Vepesid;Vepesid J;Zuyeyidal;
同义名
(-)-Etoposide;Etoposidum [INN-Latin];trans-Etoposide;
基本介绍
A semisynthetic derivative of podophyllotoxin that exhibits antitumor activity. Etoposide inhibits DNA synthesis by forming a complex with topoisomerase II and DNA. This complex induces breaks in double stranded DNA and prevents repair by topoisomerase II binding. Accumulated breaks in DNA prevent entry into the mitotic phase of cell division, and lead to cell death. Etoposide acts primarily in the G2 and S phases of the cell cycle. [PubChem]
生产厂家
- Accord healthcare inc usa
- App pharmaceuticals llc
- Bedford laboratories div ben venue laboratories inc
- Bristol myers squibb co
- Hospira inc
- Marsam pharmaceuticals llc
- Mylan pharmaceuticals inc
- Pharmachemie bv
- Pierre fabre medicament
- Teva parenteral medicines inc
- Watson laboratories inc
封装厂家
参考
| Synthesis Reference | Not Available |
| General Reference | Not Available |
剂型
| Form | Route | Strength |
|---|---|---|
| Capsule | Oral | |
| Liquid | Intravenous | |
| Solution | Intravenous |
规格
| Unit description | Cost | Unit |
|---|---|---|
| VePesid 20 50 mg capsule Box | 1273.41 USD | box |
| Etopophos 100 mg vial | 159.55 USD | vial |
| Etoposide 50 mg capsule | 47.64 USD | capsule |
| Etoposide 100 mg/5 ml vial | 28.89 USD | ml |
| Toposar 100 mg/5 ml vial | 2.25 USD | ml |
| Toposar 1000 mg/50 ml vial | 1.12 USD | ml |
| Toposar 500 mg/25 ml vial | 1.06 USD | ml |
化合物类型
| Type | small molecule |
| Classes |
|
| Substructures |
|
适应症
Cancer 癌症;
药理
| Indication | For use in combination with other chemotherapeutic agents in the treatment of refractory testicular tumors and as first line treatment in patients with small cell lung cancer. Also used to treat other malignancies such as lymphoma, non-lymphocytic leukemia, and glioblastoma multiforme. | ||||||||||
| Pharmacodynamics | Etoposide is an antineoplastic agent and an epipodophyllotoxin (a semisynthetic derivative of the podophyllotoxins). It inhibits DNA topoisomerase II, thereby ultimately inhibiting DNA synthesis. Etoposide is cell cycle dependent and phase specific, affecting mainly the S and G2 phases. Two different dose-dependent responses are seen. At high concentrations (10 µg/mL or more), lysis of cells entering mitosis is observed. At low concentrations (0.3 to 10 µg/mL), cells are inhibited from entering prophase. It does not interfere with microtubular assembly. The predominant macromolecular effect of etoposide appears to be the induction of DNA strand breaks by an interaction with DNA-topoisomerase II or the formation of free radicals. | ||||||||||
| Mechanism of action | Etoposide inhibits DNA topoisomerase II, thereby inhibiting DNA re-ligation. This causes critical errors in DNA synthesis at the premitotic stage of cell division and can lead to apoptosis of the cancer cell.. Etoposide is cell cycle dependent and phase specific, affecting mainly the S and G2 phases of cell division. | ||||||||||
| Absorption | Absorbed well, time to peak plasma concentration is 1-1.5 hrs. Mean bioavailability is 50%. | ||||||||||
| Volume of distribution | Not Available | ||||||||||
| Protein binding | 97% | ||||||||||
| Metabolism |
Primarily hepatic (through O-demethylation via the CYP450 3A4 isoenzyme pathway) with 40% excreted unchanged in the urine.
|
||||||||||
| Route of elimination | Etoposide is cleared by both renal and nonrenal processes, i.e., metabolism and biliary excretion. Glucuronide and/or sulfate conjugates of etoposide are also excreted in human urine. Biliary excretion of unchanged drug and/or metabolites is an important route of etoposide elimination as fecal recovery of radioactivity is 44% of the intravenous dose. Only 8% or less of an intravenous dose is excreted in the urine as radiolabeled metabolites of 14C-etoposide. | ||||||||||
| Half life | 4-12 hours | ||||||||||
| Clearance |
|
||||||||||
| Toxicity | Side effects include alopecia, constipation, diarrhea, nausea and vomiting and secondary malignancies (leukemia). | ||||||||||
| Affected organisms |
|
||||||||||
| Pathways |
|
理化性质
| Properties | |||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| State | solid | ||||||||||||||||||||||||||||||||||||
| Melting point | 236-251 oC | ||||||||||||||||||||||||||||||||||||
| Experimental Properties |
|
||||||||||||||||||||||||||||||||||||
| Predicted Properties |
|
||||||||||||||||||||||||||||||||||||
药物相互作用
| Drug | Interaction |
|---|---|
| Aprepitant | Aprepitant may change levels of the chemotherapy agent, etoposide. |
| Cyclosporine | Cyclosporine may increase the therapeutic and adverse effects of etoposide. |
| Quinupristin | This combination presents an increased risk of toxicity |
| Telithromycin | Telithromycin may reduce clearance of Etoposide. Consider alternate therapy or monitor for changes in the therapeutic/adverse effects of Etoposide if Telithromycin is initiated, discontinued or dose changed. |
| Trastuzumab | Trastuzumab may increase the risk of neutropenia and anemia. Monitor closely for signs and symptoms of adverse events. |
| Voriconazole | Voriconazole, a strong CYP3A4 inhibitor, may increase the serum concentration of etoposide by decreasing its metabolism. Monitor for changes in the therapeutic and adverse effects of etoposide if voriconazole is initiated, discontinued or dose changed. |
食物相互作用
- Grapefruit and grapefruit juice should be avoided throughout treatment as grapefruit can decrease serum levels of this product.
